
Synthesis of α‐Trifluoromethylated Ketones from Vinyl Triflates in the Absence of External Trifluoromethyl Sources - Kawamoto - 2017 - Angewandte Chemie International Edition - Wiley Online Library

Vinyl triflates derived from 1,3-dicarbonyl compounds and analogs: access and applications to organic synthesis - ScienceDirect

Synthesis of β-Boryl-α,β-unsaturated Carbonyl Compounds via Palladium-Catalyzed Cross-Coupling Reaction of Bis(pinacolato)diboron with Vinyl Triflates

Palladium-catalyzed conversion of aryl and vinyl triflates to bromides and chlorides. - Abstract - Europe PMC

Radical Desulfur-Fragmentation and Reconstruction of Enol Triflates: Facile Access to α-Trifluoromethyl Ketones. | Semantic Scholar

Vinyl Triflate–Aldehyde Reductive Coupling–Redox Isomerization Mediated by Formate: Rhodium‐Catalyzed Ketone Synthesis in the Absence of Stoichiometric Metals - Shuler - 2019 - Chemistry – A European Journal - Wiley Online Library

Nickel-Catalyzed Cross-Electrophile Vinyl–Vinyl Coupling: An Approach to Structurally Versatile Dienylboronates | CCS Chem

Palladium‐Catalyzed Fluorination of Cyclic Vinyl Triflates: Effect of TESCF3 as an Additive - Ye - 2016 - Angewandte Chemie International Edition - Wiley Online Library

Palladium-catalysed coupling of vinyl triflates with enynes and its application to the synthesis of 1α,25-dihydroxyvitamin D3 - ScienceDirect

Vinyl Triflate–Aldehyde Reductive Coupling–Redox Isomerization Mediated by Formate: Rhodium‐Catalyzed Ketone Synthesis in the Absence of Stoichiometric Metals - Shuler - 2019 - Chemistry – A European Journal - Wiley Online Library

Vinyl Triflate–Aldehyde Reductive Coupling–Redox Isomerization Mediated by Formate: Rhodium‐Catalyzed Ketone Synthesis in the Absence of Stoichiometric Metals - Shuler - 2019 - Chemistry – A European Journal - Wiley Online Library
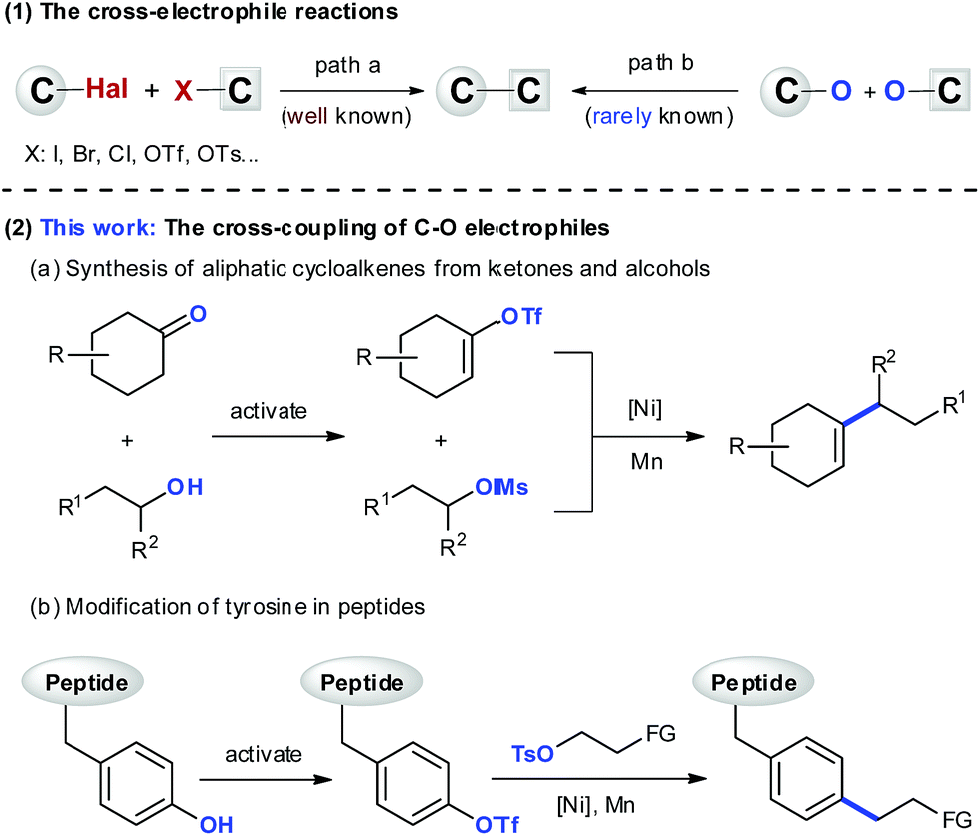
Ni-catalyzed cross-electrophile coupling between vinyl/aryl and alkyl sulfonates: synthesis of cycloalkenes and modification of peptides - Chemical Science (RSC Publishing) DOI:10.1039/C9SC03347E

PDF) Zinc triflate-mediated cyclopropanation of oxindoles with vinyl diphenyl sulfonium triflate: a mild reaction with broad functional group compatibility

Vinyl triflates derived from 1,3-dicarbonyl compounds and analogs: access and applications to organic synthesis - ScienceDirect

Vinyl triflates derived from 1,3-dicarbonyl compounds and analogs: access and applications to organic synthesis - ScienceDirect

A Versatile and Highly Stereoselective Access to Vinyl Triflates Derived from 1,3-Dicarbonyl and Related Compounds







