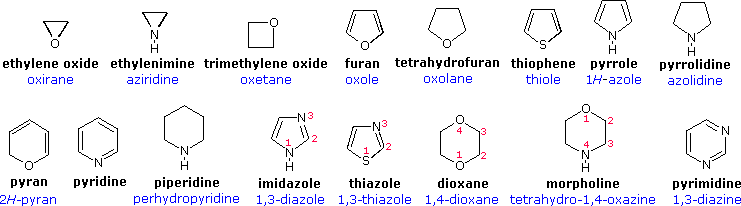
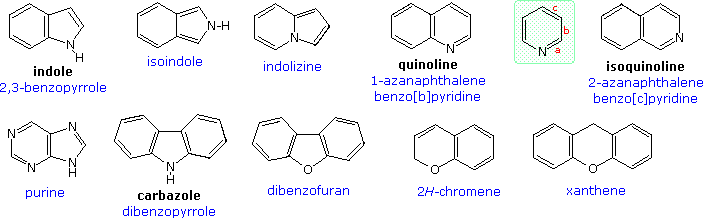
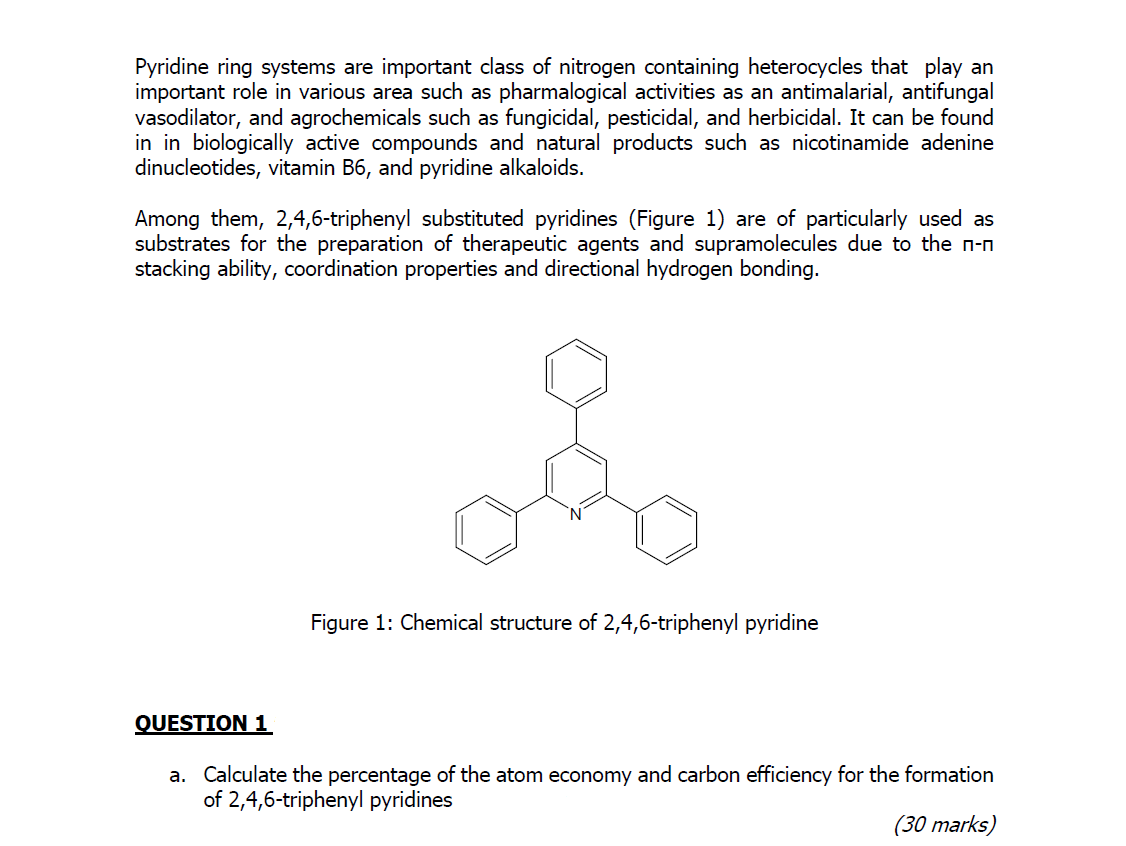
![Crystal Correlation Of Heterocyclic Imidazo[1,2-a]pyridine Analogues and Their Anticholinesterase Potential Evaluation | Scientific Reports Crystal Correlation Of Heterocyclic Imidazo[1,2-a]pyridine Analogues and Their Anticholinesterase Potential Evaluation | Scientific Reports](https://media.springernature.com/m685/springer-static/image/art%3A10.1038%2Fs41598-018-37486-7/MediaObjects/41598_2018_37486_Fig1_HTML.png)
Crystal Correlation Of Heterocyclic Imidazo[1,2-a]pyridine Analogues and Their Anticholinesterase Potential Evaluation | Scientific Reports

Polarization of the Pyridine Ring: Highly Functionalized Piperidines from Tungsten−Pyridine Complex | Journal of the American Chemical Society

Investigations into the Mechanisms of Pyridine Ring Cleavage in Vismodegib | Drug Metabolism & Disposition

Pyrrole ring opening – pyridine ring closure: Recyclization of 2-(2-oxo-1,2-dihydro-3H-pyrrol-3-ylidene)malononitriles into highly functionalized nicotinonitriles - ScienceDirect

Pyridine Ring Stock Illustrations – 3 Pyridine Ring Stock Illustrations, Vectors & Clipart - Dreamstime

Pyridine is a benzene ring with one of the carbon atoms substituted with nitrogen. What are the hybridizations of atoms 1 and 2 respectively in the structure of pyridine? | Homework.Study.com
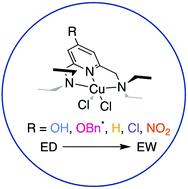
Electronic influence of substitution on the pyridine ring within NNN pincer-type molecules - Dalton Transactions (RSC Publishing)
![The [1,2,3]Triazolo[1,5-a]pyridine ring: A sensitive sensor for the electronic profile of phosphorus substituents - Dalton Transactions (RSC Publishing) The [1,2,3]Triazolo[1,5-a]pyridine ring: A sensitive sensor for the electronic profile of phosphorus substituents - Dalton Transactions (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/B906034K)
The [1,2,3]Triazolo[1,5-a]pyridine ring: A sensitive sensor for the electronic profile of phosphorus substituents - Dalton Transactions (RSC Publishing)
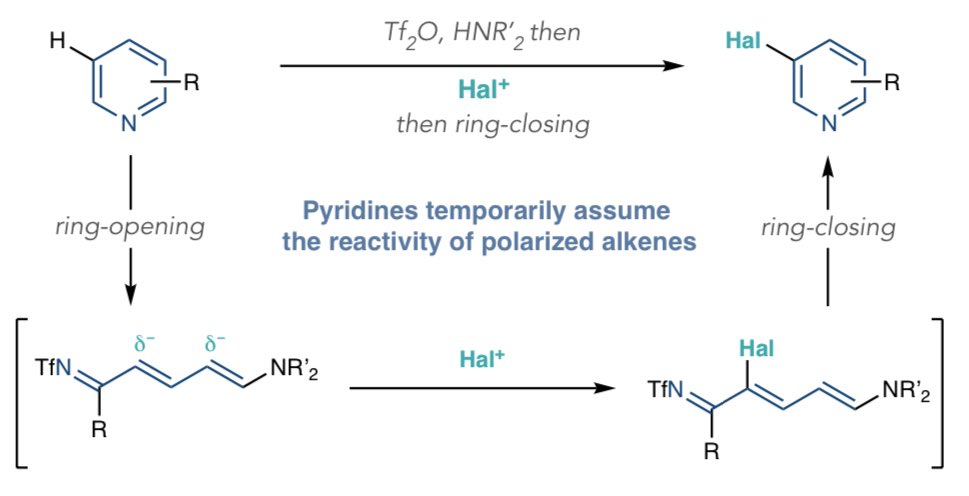
Procter Group on Twitter: "#ProcterPOW: The groups of @AndyMcNally367 and @bobbypaton describe an ingenious 3-selective halogenation of pyridines that proceeds by ring-opening🔓, electrophilic halogenation of Zincke imine intermediates, and ring ...