Balanced the following reactions:1. Cu + H2SO4→ CuSO4 + SO2↑ + H2O 2. Fe + H2O→Fe3O4 + H2↑ 3. CH4 + O2→ CO2+H2O

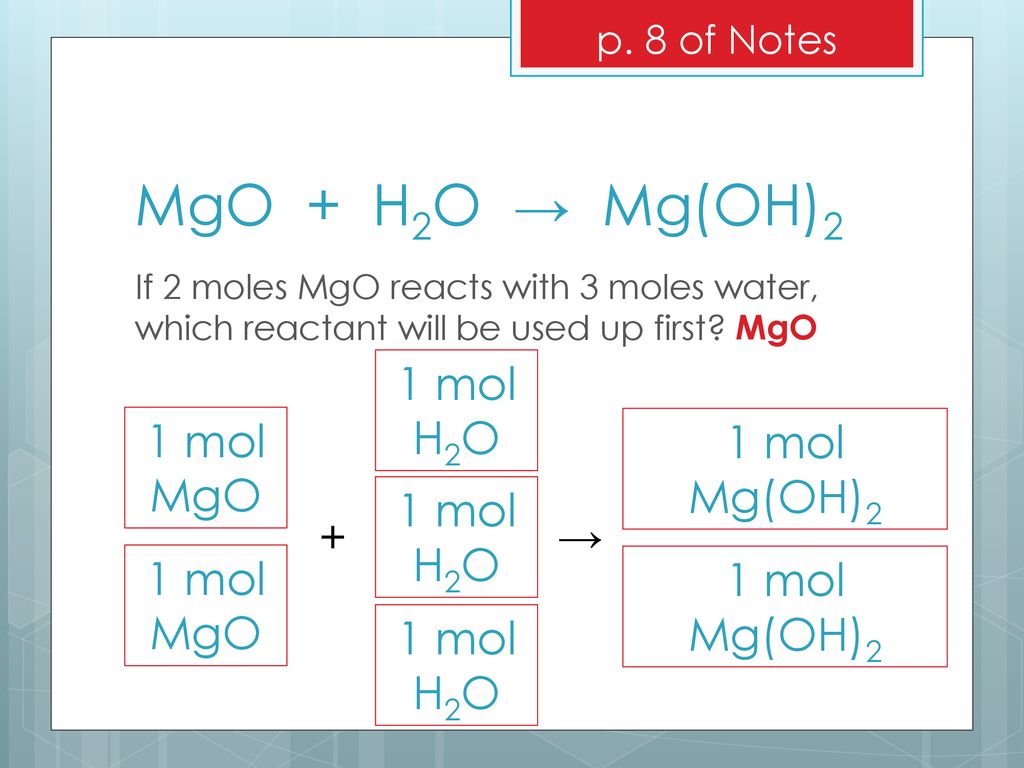
E 21 (1 Point) Which of the following represent the reaction between MgO and H2O? a)... - HomeworkLib
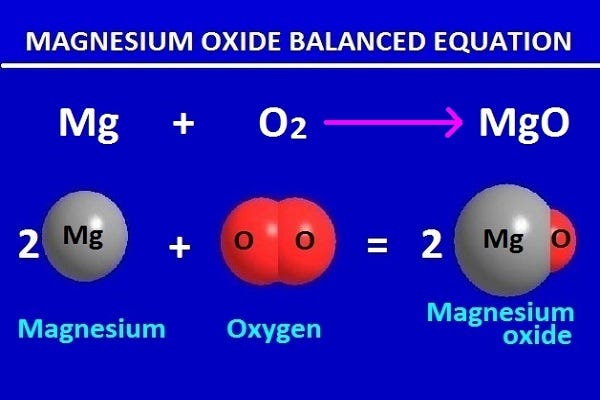
Magnesium oxide balanced equation in chemistry for class 9 | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium
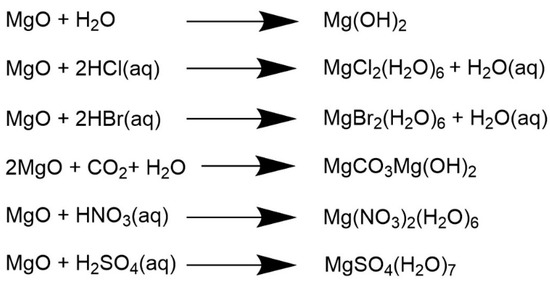
Molecules | Free Full-Text | Computational and Experimental 1H-NMR Study of Hydrated Mg-Based Minerals | HTML

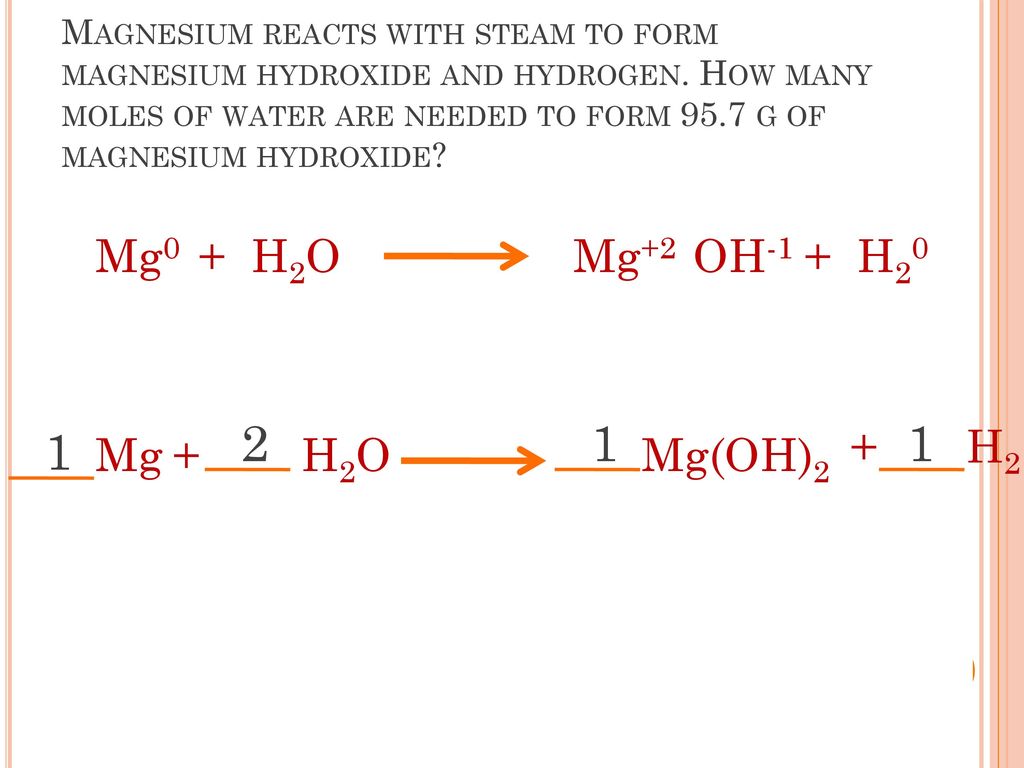
Balance the following equations.(i) KMnO2 + HCl → KCl + MnCl2 + H2 O + Cl2 (ii) NH3 + O3 → NO + H2 O
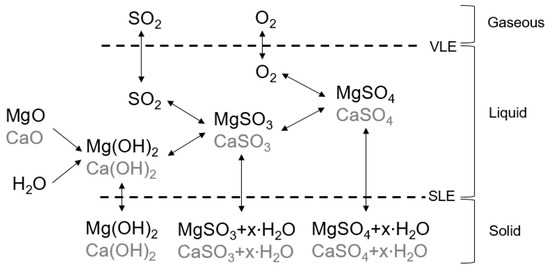
Processes | Free Full-Text | Solubility Data of Potential Salts in the MgO -CaO-SO2-H2O-O2 System for Process Modeling | HTML



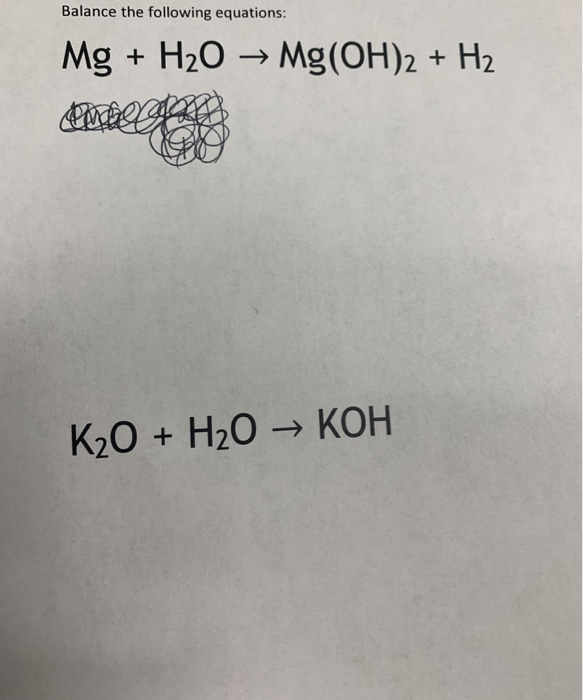




.PNG)