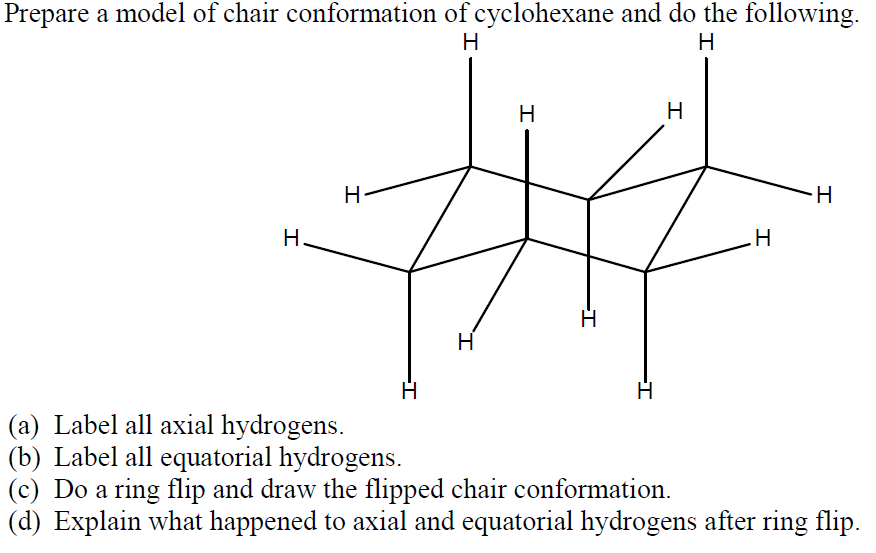
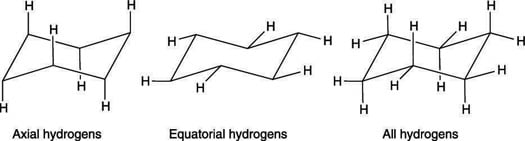
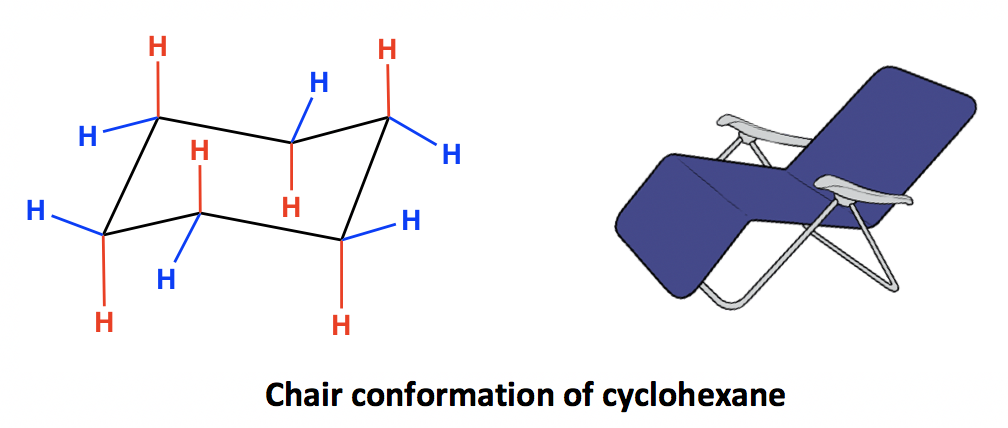
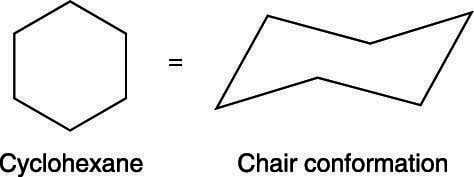
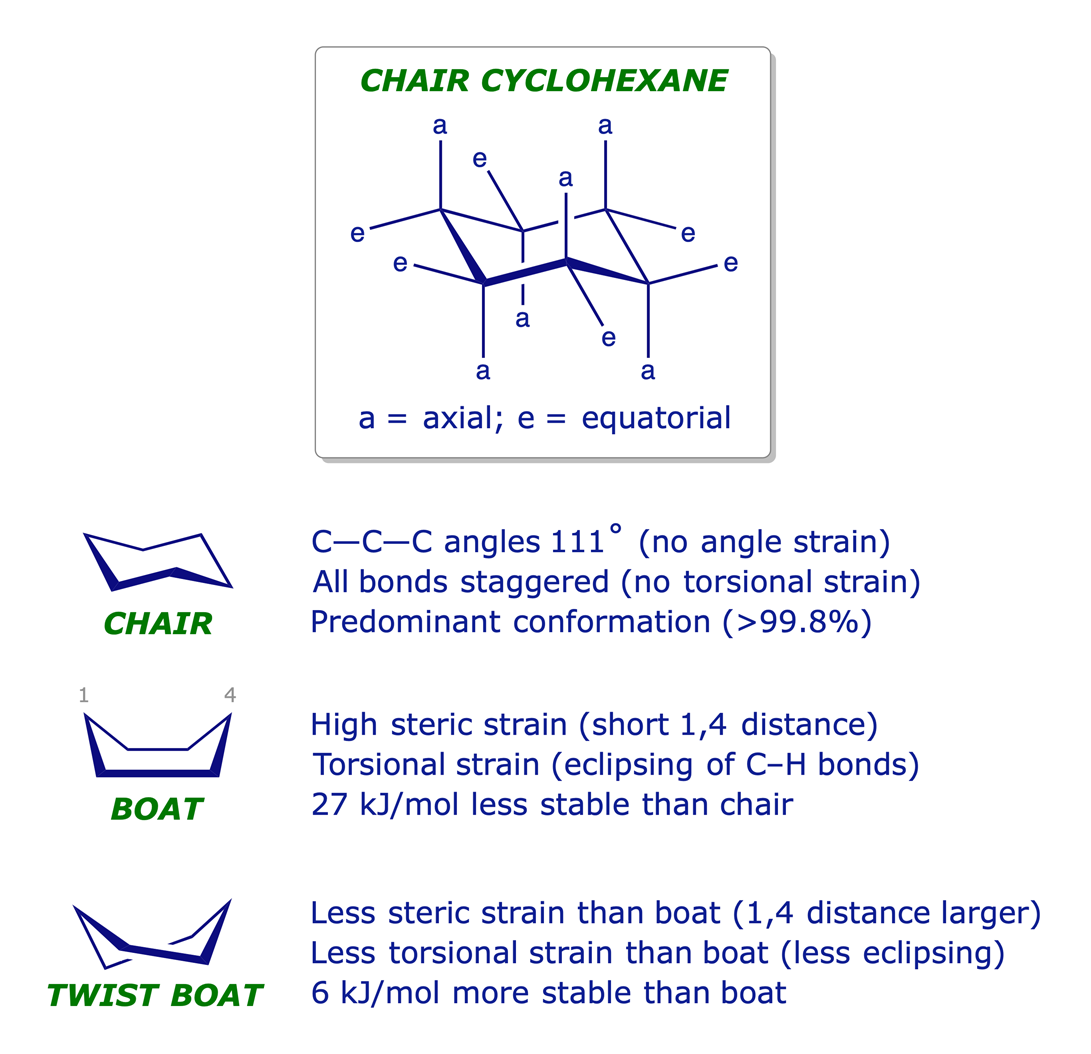
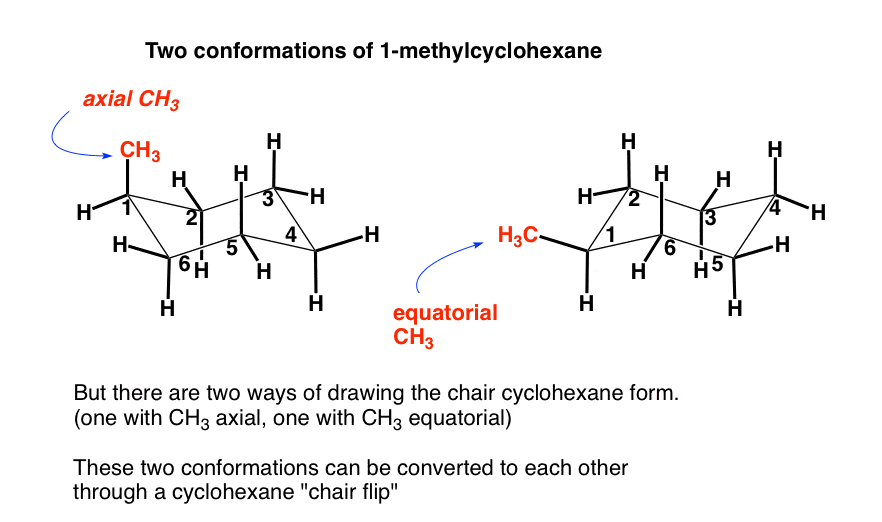
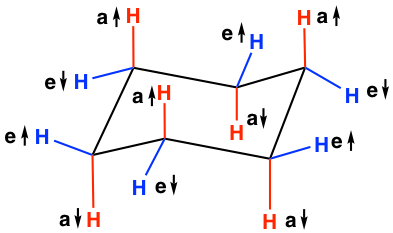
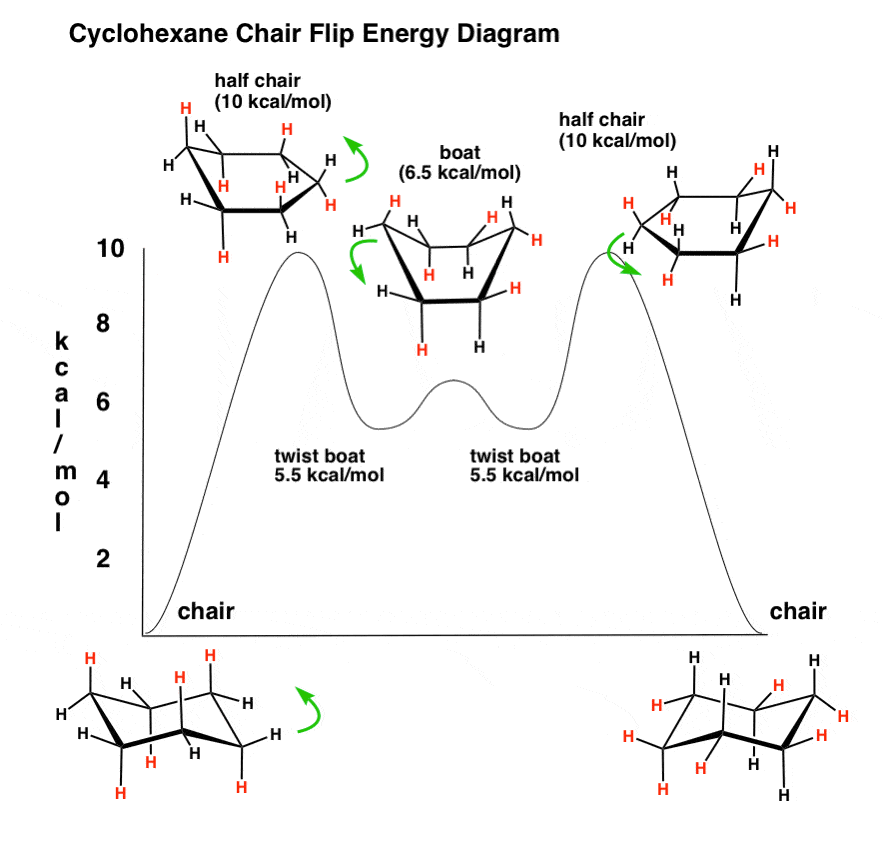
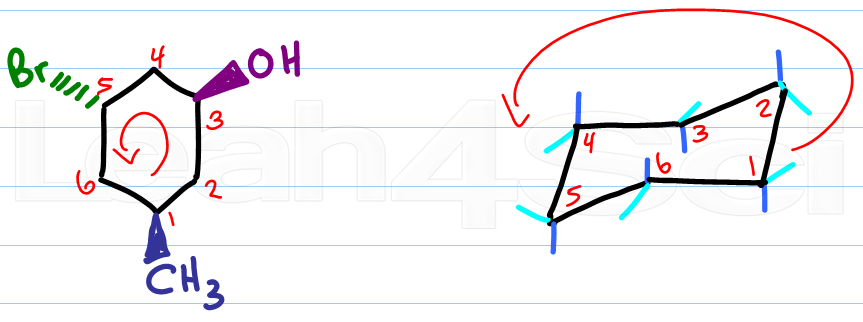
Given cyclohexane in a chair conformation, substitute two of the H labels with bromine and chlorine to construct the more stable conformation of cis-1-bromo-3-chlorocyclohexane. Use the numbering prov | Study.com

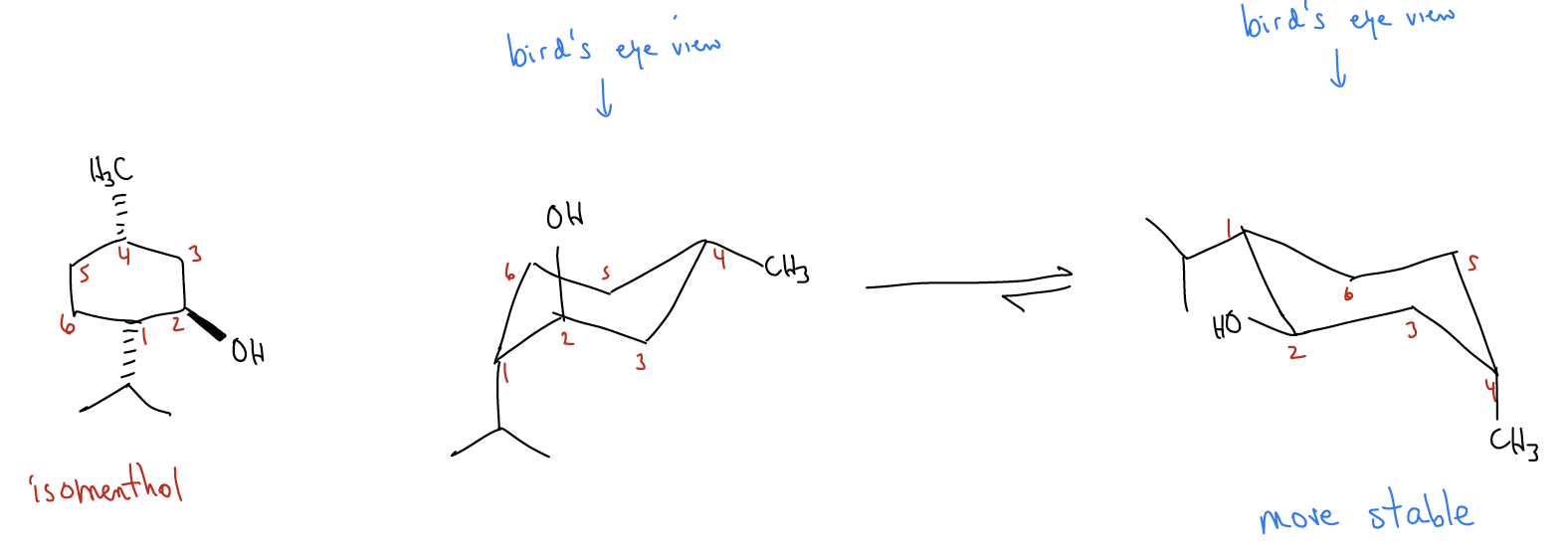
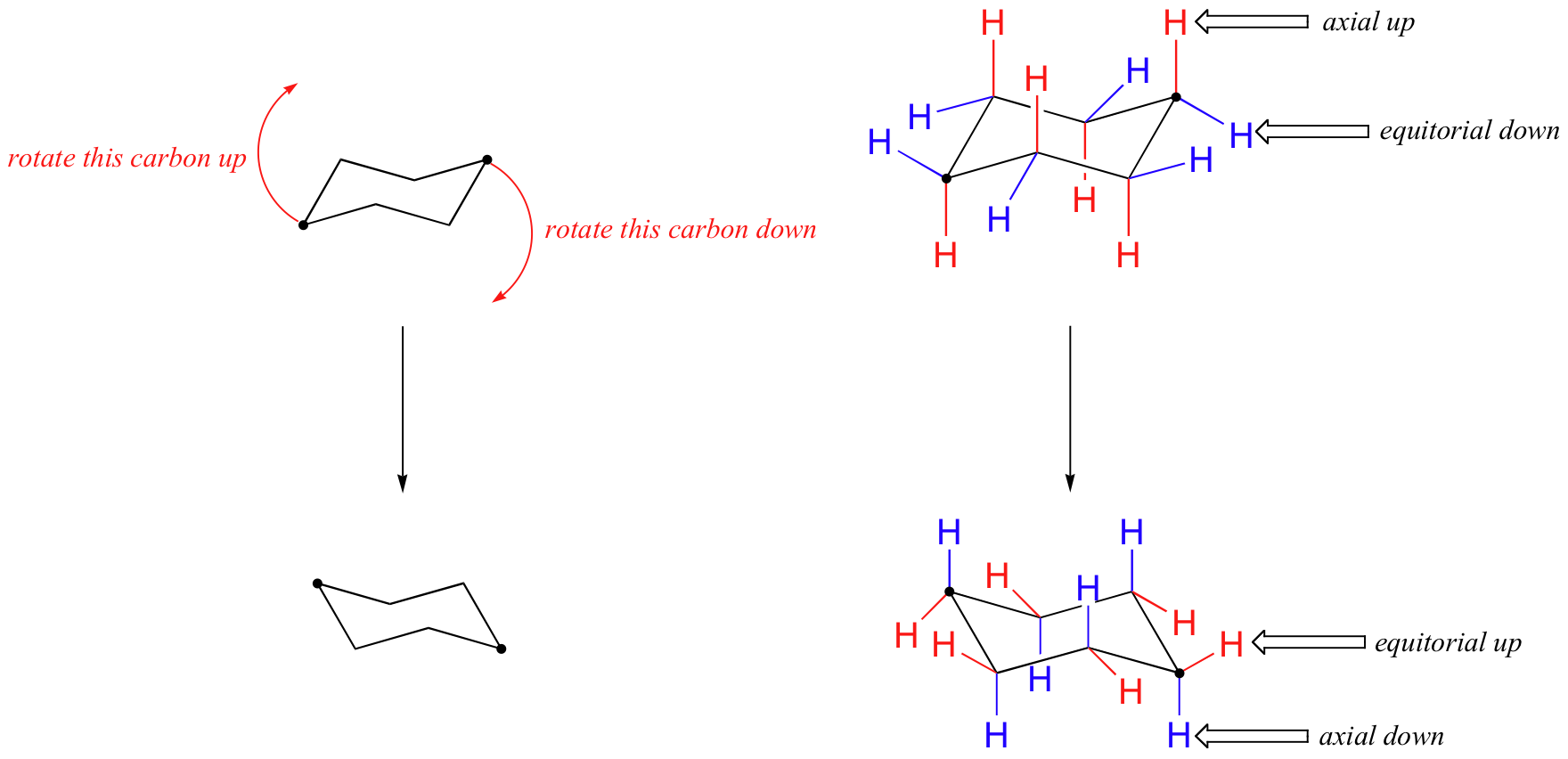
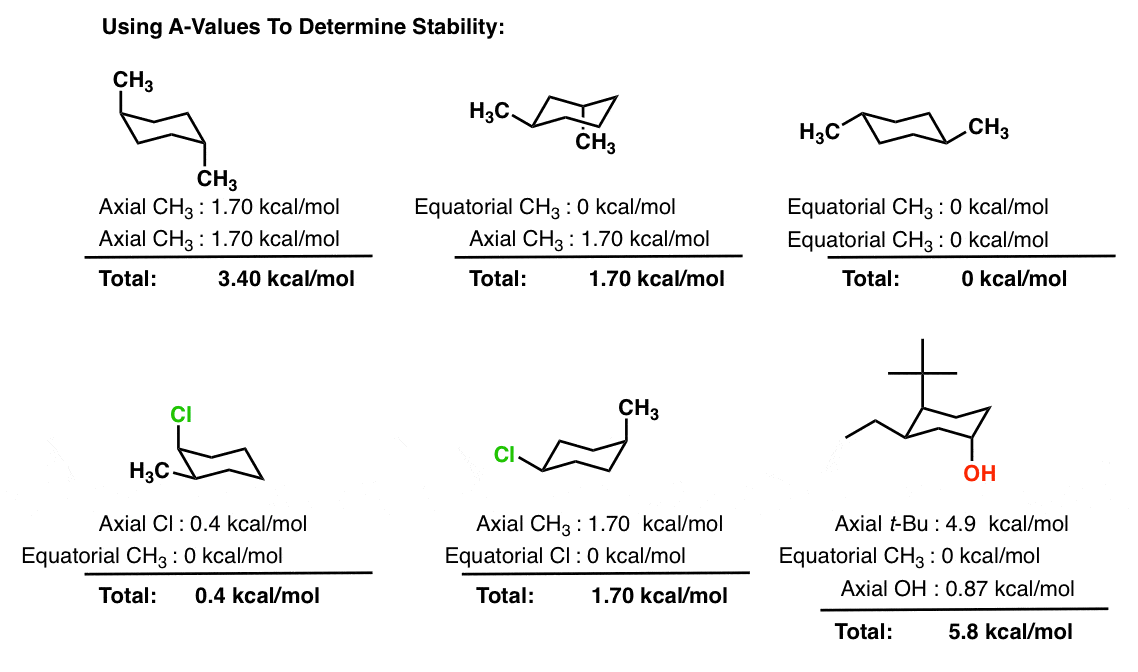
Stereoisomeric Conformations, Different Perspectives, Achiral Stereoisomers and Meso Compounds | Organic Chemistry Solutions