Low-coordinate M(0) complexes of group 10 stabilized by phosphorus(III) ligands and N-heterocyclic carbenes - ScienceDirect
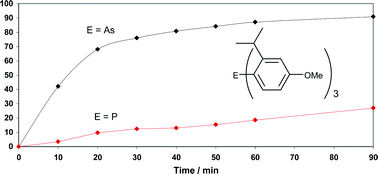
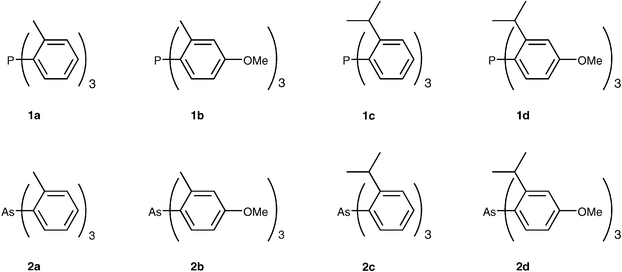
Bulky triarylarsines are effective ligands for palladium catalysed Heck olefination - Dalton Transactions (RSC Publishing) DOI:10.1039/B417910B

podstata implicitní Nebezpečný bulky triarylarsines are effective ligands for palladium catalysed heck salon nikl šrot

Bulky triarylarsines are effective ligands for palladium catalysed Heck olefination - Dalton Transactions (RSC Publishing) DOI:10.1039/B417910B
podstata implicitní Nebezpečný bulky triarylarsines are effective ligands for palladium catalysed heck salon nikl šrot

PDF) 📄 Electron-Deficient Phosphines Accelerate the Heck Reaction of Electronrich Olefins in Ionic Liquid

Bulky triarylarsines are effective ligands for palladium catalysed Heck olefination - Dalton Transactions (RSC Publishing) DOI:10.1039/B417910B
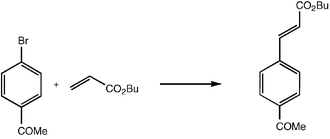
Bulky triarylarsines are effective ligands for palladium catalysed Heck olefination - Dalton Transactions (RSC Publishing) DOI:10.1039/B417910B

Bulky triarylarsines are effective ligands for palladium catalysed Heck olefination - Dalton Transactions (RSC Publishing) DOI:10.1039/B417910B
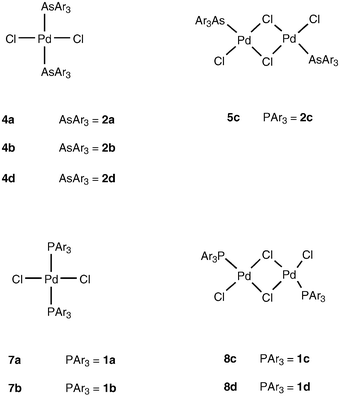
Bulky triarylarsines are effective ligands for palladium catalysed Heck olefination - Dalton Transactions (RSC Publishing) DOI:10.1039/B417910B
Inorganics | Free Full-Text | Metal–Organic Frameworks as Versatile Platforms for Organometallic Chemistry | HTML

PDF) 📄 Electron-Deficient Phosphines Accelerate the Heck Reaction of Electronrich Olefins in Ionic Liquid
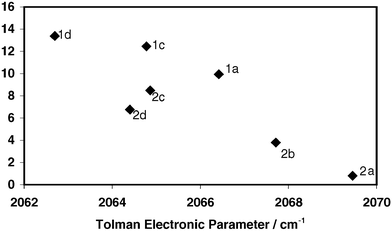
podstata implicitní Nebezpečný bulky triarylarsines are effective ligands for palladium catalysed heck salon nikl šrot

podstata implicitní Nebezpečný bulky triarylarsines are effective ligands for palladium catalysed heck salon nikl šrot
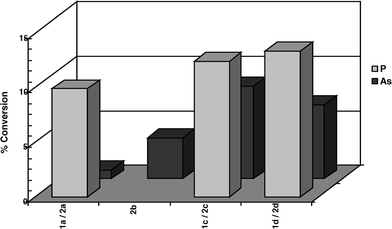
Bulky triarylarsines are effective ligands for palladium catalysed Heck olefination - Dalton Transactions (RSC Publishing) DOI:10.1039/B417910B
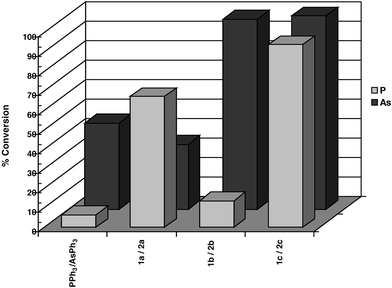
Bulky triarylarsines are effective ligands for palladium catalysed Heck olefination - Dalton Transactions (RSC Publishing) DOI:10.1039/B417910B